Our Research
Our interests span multiple neurodegenerative diseases, while focusing on the 17q21.31 locus and tau
The role of ELAVL4 expression in early synaptic dysfunction

Using forebrain organoids carrying FTD-causing MAPT mutations, we previously demonstrated that increased expression of the RNA binding protein ELAVL4 lead to alterations in glutamate synaptic signaling, making neurons more susceptible to death. Following on from this work, we are now investigating the mechanism by which ELAVL4 and mutant tau interact to disrupt the synapse, and whether we can reverse this process in order to protect neurons.
How does astrocyte function modulate neuronal health in primary tauopathies?
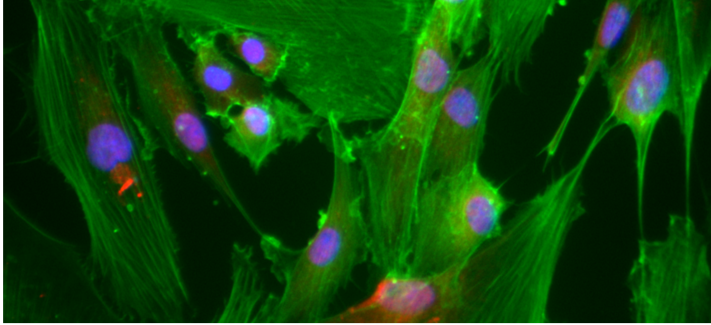
MAPT expression is highest in neurons, but it is present at very low level in astrocytes. Furthermore, many primary tauopathies are characterised by the accumulation of tau in astrocytes. This project aims to investigate the effect that MAPT expression and accumulation in astrocytes has both on the astrocytes themselves (cell autonomous effects) and on surrounding neurons (non-cell autonomous effects).
What is the functional impact of increased 4R tau?
The expression of 4R tau is crucial to adult human brain function, but this isoform specifically accumulates in several primary tauopathies, such as PSP. However, iPSC-neurons typically do not express much 4R tau, so it has been challenging to investigate the role of this isoform in these models. We have generated a unique set of isogenic iPSC lines with MAPT mutations that cause high levels of 4R tau in iPSC-neurons, and we are now characterising the effect of these mutations in 2D and 3D culture models, and in human brain.
How does LRRC37A2 protect against Parkinson's?

We have previously found that higher LRRC37A2 expression may be protective against risk for Parkinson's, but we don't know why. It is an interesting gene in the 17q21.31 "MAPT" locus with multiple copy number variants that has not been previously studied. We are using iPSC models to try to understand what the function of LRRC37A2 is in astrocytes, and how increasing its expression may protect neurons.
How does mutant MAPT affect mitochondrial function?
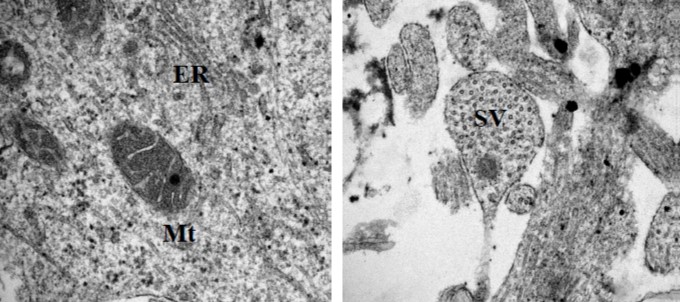
Differences in mitochondrial metabolism and oxidative phosphorylation have been observed across multiple different models of tauopathy, but it is not clear how tau or mutant tau might interact with mitochondria to cause them to behave differently. We are using iPSC-organoid models spanning different MAPT mutations to investigate different aspects of mitochondrial health, and how this changes with disease progression.





